Author: Frances Verter, PhD
During the spring 2020 pandemic lockdown, we launched a series of blogs about the use of mesenchymal stromal cells (MSC) to treat respiratory complications of COVID-19. Unfortunately, the clinical trials run prior to COVID-19 were not able to demonstrate that MSC have efficacy, against controls, at healing lung inflammation. This was addressed in our blog of 2020-04-26, The Role of MSC to Treat Coronavirus Pneumonia and ARDS. Part 1: Is the Emperor Wearing Clothes?, as well as our blog of 2020-05-25, A Role for MSC to Treat Coronavirus Patients. Part 2: Fibrosis in Survivors.
Now we are revisiting this topic with an optimistic story about the bone marrow MSC product Remestemcel-L (RYONCILTM) from Mesoblast. It turns out that Mesoblast has an edge in the battle against COVID-19 that has nothing to do with MSC trials for lung disorders: success treating GvHD1. Below we explain why this may be relevant for treating COVID-19.
Graft versus Host Disease (GvHD) is a common complication of stem cell transplants, afflicting up to half of patients each year2. The simplistic explanation of GvHD is that the cells in the graft recognize the host body as foreign and attack it. The biological mechanism by which the cells communicate is to secrete signaling proteins called cytokines3. In fact, the levels of specific cytokines can be used as part of a panel of biomarkers to predict GvHD outcomes4. Among pediatric patients with steroid-resistant GvHD, the survival rate is only 20%5,6. Hence, MSC cell-based therapy has been studied as a potential method to suppress the inflammation and immune response associated with GvHD.
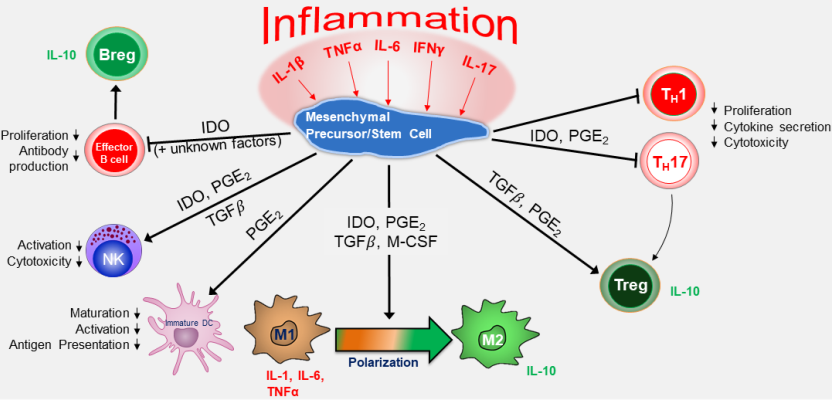
On 13 August 2020, the U.S. FDA agreed to review the Mesoblast MSC product Remestemcel-L for a Biologics License to treat pediatric GvHD1,7,8. Mesoblast has run three phase 3 trials in which they achieved 65-75% overall survival of children with steroid-resistant GvHD, more than triple the previous survival rate8. Figure 1 is an infographic from Mesoblast, reproduced with permission, which describes the mechanism of action by which MSC are believed to dampen the immune and inflammatory response of GvHD. The five cytokine markers that Mesoblast considers critical are interferon gamma (IFN-γ), interleukin 1beta, (IL-1β), interleukin 6 and 17 (IL-6, IL-17), and tumor necrosis factor alpha (TNF-α)1.
The novel coronavirus COVID-19 is also an illness characterized by dangerous levels of inflammation and an intense immune reaction that can lead to a storm of cytokines9. On 7 May 2020, Cytotherapy published a very interesting paper that looked at the overlap between COVID-19 versus other syndromes that involve inflammation and immune dysfunction10. The authors focused on syndromes that occur in the context of cellular therapies. They compared COVID-19 against clinical and laboratory characteristics of three syndromes: Cytokine Release Syndrome (CRS), Immune Reconstitution Inflammatory Syndrome (IRIS), and Secondary Hemophagocytic Lymphohistiocytosis (sHLH). Figure 2, shown with the authors’ permission, is a slightly modified version of their Venn diagram displaying the overlap between the cytokine markers of COVID-19 versus these three inflammatory conditions.
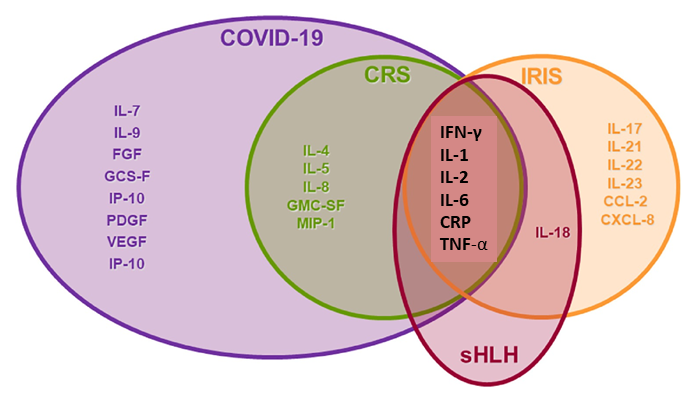
The pattern of cytokine response seen in COVID-19 is meaningful for the clinical management and outcome of the disease. On 24 August 2020, a paper was published in Nature Medicine which argued that the inflammatory cytokine signature in COVID-19 patients is predictive of severity of illness and survival11,12. This conclusion is based on analysis of 1484 patients hospitalized at Mount Sinai Health System, plus a validation cohort of 231 patients11. The authors reported that levels of interleukin-6 (IL-6) and tumor necrosis factor alpha (TNF-α) predicted both COVID-19 severity and death, after adjusting for other laboratory inflammation markers, disease severity, patient ages and comorbidities.
To date, no one has compared the cytokine patterns of COVID-19 versus GvHD. We decided to make the first attempt at this, shown in Figure 3. We reproduced the format of the figure in Cancio et al.10, and adopted their list of COVID-19 cytokines, which is based on Herold et al.9. For GvHD, we selected a panel of cytokines which were frequently cited in review articles about GvHD3-5,13,14. We must caution that this diagram is only suggestive, because we did not conduct a comprehensive literature review, thus we do not have a definitive list of the leading cytokines associated with GvHD. Nonetheless, we can demonstrate that there is a short list of cytokines associated with inflammation that is definitely common to both COVID-19 and GvHD.
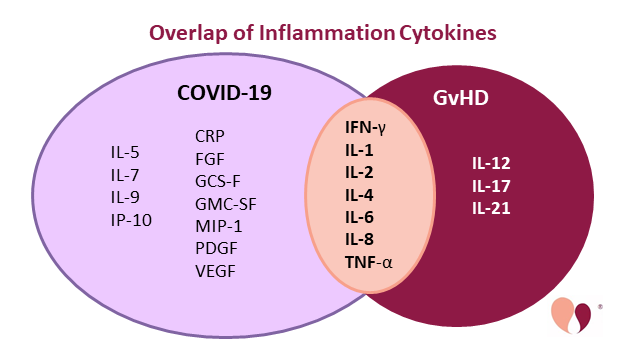
Based on the overlapping cytokine signature between COVID-19 and GvHD, plus the evidence that these signatures have prognostic significance, it is not unreasonable to hope that the Remestemcel-L MSC product that can suppress GvHD may also have benefit for COVID-19 patients. Mesoblast is currently running a clinical trial NCT04371393 for COVID-19-induced ARDS that started at Mount Sinai hospital but has expanded to 17 locations and hopes to recruit 300 patients.
Also of note, children infected with COVID-19 sometimes develop a multisystem inflammatory syndrome (MIS-C). This is a newly identified diagnosis, and early reports indicate that the cytokine profile of MIS-C is unique, even compared to severe COVID-1915. Mesoblast is running an expanded access program NCT04456439 for MIS-C.
CellTrials.org has identified 85 clinical trials which employ MSC to treat COVID-19 that were registered during the first half of 202016. However, only Mesoblast and Athersys are running MSC trials that aim to recruit hundreds of patients. A success in any of these trials will be great news for developers of MSC products and for cell therapy in general, plus of course for COVID-19 patients and their families.
References:
- 1. Mesoblast. Remestemcel-L for treatment of steroid refractory acute graft versus host disease in pediatric patients. Briefing Document 2020-08-13
- 2. Jacobsohn DA, and Vogelsang GB. Acute graft versus host disease. Orphanet J Rare Diseases 2007; 2:35.
- 3. Zeiser R, Blazar BR. Pathophysiology of Chronic Graft-versus-Host Disease and Therapeutic Targets. NEJM 2017; 377:2565-79.
- 4. Levine JE, Logan BR, Wu J, et al. Acute graft-versus-host disease biomarkers measured during therapy can predict treatment outcomes: a Blood and Marrow Transplant Clinical Trials Network study. Blood 2012; 119:3854-60.
- 5. MacMillan ML, Robin M, Harris AC, et al. A Refined Risk Score for Acute Graft-versus-Host Disease that Predicts Response to Initial Therapy, Survival, and Transplant-Related Mortality. Biol. Blood and Marrow Transpl. 2015; 21(4):761-767.
- 6. Malard F, Huang XJ, Sim JPY. Treatment and unmet needs in steroid-refractory acute graft-versus-host disease. Leukemia 2020; 34:1229–1240.
- 7. FDA. Oncologic Drugs Advisory Committee (ODAC) Meeting. Briefing Document 2020-08-13
- 8. Ricki Lewis. Mesoblast MSCs Quell Peds GvHD; On Road to FDA Approval? THE NICHE Knoepfler Lab Stem Cell Blog 2020-08-20
- 9. Herold T, Jurinovic V, Arnreich C, et al. Elevated levels of IL-6 and CRP predict the need for mechanical ventilation in COVID-19. J. Allergy and Clinical Immun. 2020; 146(1):128-136.
- 10. Cancio M, Ciccocioppo R, Rocco P, … Hanley PJ. Emerging trends in COVID-19 treatment: learning from inflammatory conditions associated with cellular therapies. Cytotherapy 2020; 22(9):474-481.
- 11. Del Valle DM, Kim-Schulze S, ... Gnjatic S. An inflammatory cytokine signature predicts COVID-19 severity and survival. Nature Medicine 2020-08-24;
- 12. Walker M. Could COVID-19 'Inflammatory Signature' Predict Disease Course? MedPage Today 2020-08-25
- 13. Coghill JM, Sarantopoulos S, Moran TP, et al. Effector CD4+ T cells, the cytokines they generate, and GVHD: something old and something new. Blood 2011; 117(12):3268–3276.
- 14. Henden AS, and Hill GR. Cytokines in Graft-versus-Host Disease. Journal Immunology 2015; 194(10):4604-4612.
- 15. Diorio C, ... Teachey DT, Bassiri H. Multisystem inflammatory syndrome in children and COVID-19 are distinct presentations of SARS-CoV-2. J Clinical Investigation in press doi 10.1172/JCI140970
- 16. Verter F. and Couto PS. Development of COVID-19 therapies from birthing tissues and cord blood. Stem Cells Translational Medicine Supplement. Cord Blood Connect: The International Congress for Cord Blood and Perinatal Tissue Research 2020. 2020; 9(S1):S15.