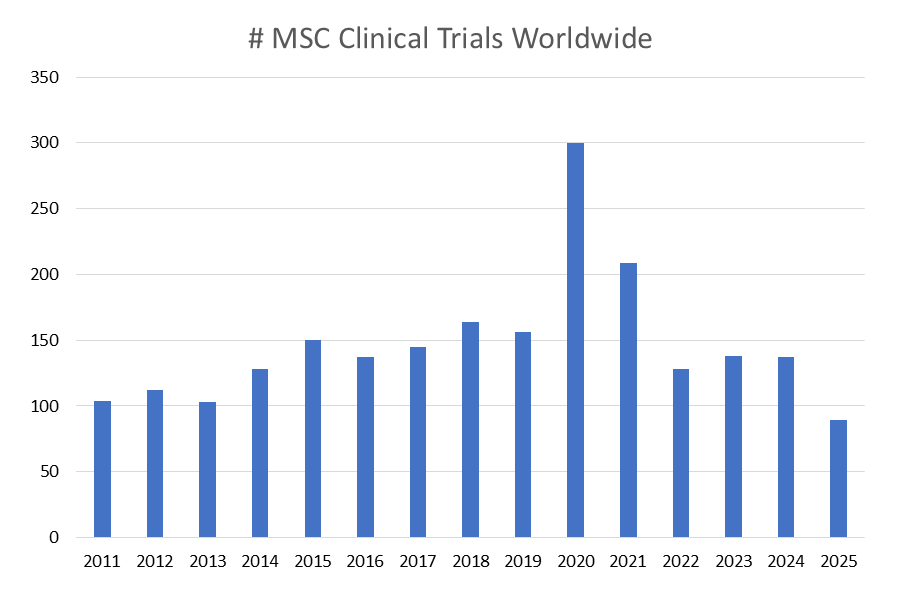
We have been compiling MSC clinical trials since 2011. For the past decade, a typical year holds around 150 MSC trials. This number doubled in 2020, due to the huge number of MSC trials treating COVID-19. For this database we are offering clients MSC trials data from the past five years. Within the past couple of years, nearly half of MSC trials are NOT registered in ClinicalTrials.gov: 47% were outside in 2024 and 48% are outside in the first half of 2025.
These trials employ isolated Mesenchymal Stromal Cells (MSC). We do not include trials with bone marrow BMAC or adipose SVF, those are in our "all trials" databases. The MSC trials have been compiled from 15+ international registries.
Excel File Columns
- Trial Data
- Registration Month
- Registration Year
- Trial ID with link to trial on-line
- Country
- Phase
- Status (at registration)
- Cell Data
- Type of therapy: Immunotherapy or Regenerative
- Cell Type
- Cell Source
- Route of Administration
- Cell Dose
- Patient Data
- Indication
- Donor Autologous or Allogeneic
- Target Enrollment
- Patient Age Groups
- Sponsor Data
- Academia or Industry Funding
- Sponsor Name and any Collaborators
- Alternate Trial ID (if any)
- Product Name (if any)
| Attachment | Size |
|---|---|
| SAMPLE MSC Clinical Trials Jan. 2021 - June 2025 | 77.3 KB |