Pedro Silva Couto MSc, and Frances Verter PhD
When compiling clinical trials, it is difficult to find the best combination of keywords that will collect a complete set of relevant trials. We argue that counting keyword hits alone is never sufficient, that a scientist has to look at each trial returned by the search.
The first installment of this series was about compiling cell therapy trials that use cord blood. We found that among the trials in ClinicalTrials.gov that contain the keywords “cord blood” and are currently recruiting, 22% are obstetrics studies that use cord blood to measure maternal-fetal health, 31% are traditional hematology/oncology cell therapy, 26% are advanced cell therapy, and 21% are other studies or miscellaneous false positives. Thus if people searching for cord blood cell therapy relied solely on counting hits for the keywords “cord blood”, they would over estimate the number of active cell therapy trials by about a factor of two.
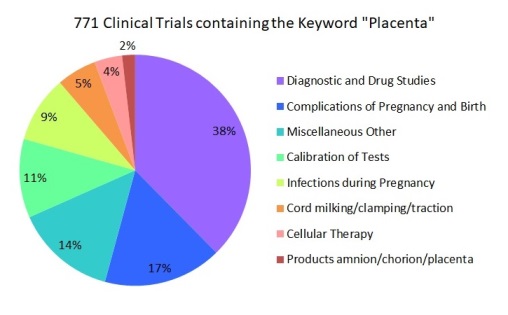 This second installment is about searching for clinical trials that are performing regenerative medicine with cells from the placenta. We searched in ClinicalTrials.gov on 10 May 2017 for all trials that contained the keyword “placenta”. We found 771 placenta trials, but only 30 of them (4%) are performing cell therapy.
This second installment is about searching for clinical trials that are performing regenerative medicine with cells from the placenta. We searched in ClinicalTrials.gov on 10 May 2017 for all trials that contained the keyword “placenta”. We found 771 placenta trials, but only 30 of them (4%) are performing cell therapy.
It is probably not surprising that a large fraction of clinical trials that involve the placenta are studies of maternal-fetal interactions during pregnancy. However, we were surprised to find that the largest group, almost half of the trials (49%), are studies that use the placenta for test calibration, as a diagnostic tool, or to measure drug efficacy. There are more placenta trials studying malaria in pregnancy (7%) than there are performing cell therapy with the placenta (4%). Several trials of cord blood transplants include the keyword placenta, but only in the sense that cord blood is harvested from the umbilical cord and placenta, so these keyword counts were false positives that had to be removed from the cell therapy total.
At this point you might think that a better way to compile clinical trials using placenta cells for therapy would be to search for all trials that have both the keywords “placenta” AND “stem cell”. But you would be wrong. The phrase “stem cell” does not always appear in cell therapy trials. The cells providing the mechanism of action might be some combination of mesenchymal cells, stromal cells, or epithelial cells. The most efficient keyword combination would be “placenta” AND “cell”, but there is still no guarantee that the word cell will appear in relevant trials. To make matters worse, the keyword “placenta” does not always appear in trials that harvest cells from the placenta!
.jpg) At the same time that we were working on this search for “placenta” trials, we were also conducting a search for clinical trials of commercial products manufactured from the amniotic membrane of the placenta (AMP). We discovered that when corporations sponsor trials of products made from the placenta, the trial name and trial description only state the name of the product, and never mention the keyword “placenta”. Even products that are explicitly claimed to hold live placenta cells, such as Grafix, do not include the word “placenta” in the description of the trial. Hence trials of products manufactured from the placenta are invisible to searches on the keyword “placenta”, and the only way to find them is to start out with a cheat sheet that lists the names of all the AMP products, which we had courtesy of Russ Schweizer.
At the same time that we were working on this search for “placenta” trials, we were also conducting a search for clinical trials of commercial products manufactured from the amniotic membrane of the placenta (AMP). We discovered that when corporations sponsor trials of products made from the placenta, the trial name and trial description only state the name of the product, and never mention the keyword “placenta”. Even products that are explicitly claimed to hold live placenta cells, such as Grafix, do not include the word “placenta” in the description of the trial. Hence trials of products manufactured from the placenta are invisible to searches on the keyword “placenta”, and the only way to find them is to start out with a cheat sheet that lists the names of all the AMP products, which we had courtesy of Russ Schweizer.
We eventually found 61 trials of AMP products, wrote a news story about the AMP trials, and posted a free download of AMP trials. These 61 trials all somehow use placenta cells and yet none of them were found in our original sweep of 771 trials with the keyword “placenta”. As a result, our new total is 832 clinical trials that use the placenta cells, 4% of them in cellular therapy and 9% as part of a manufactured product. So far as counting keywords, there are 8% more clinical trials using placenta cells than can be found by searching on the keyword “placenta”.